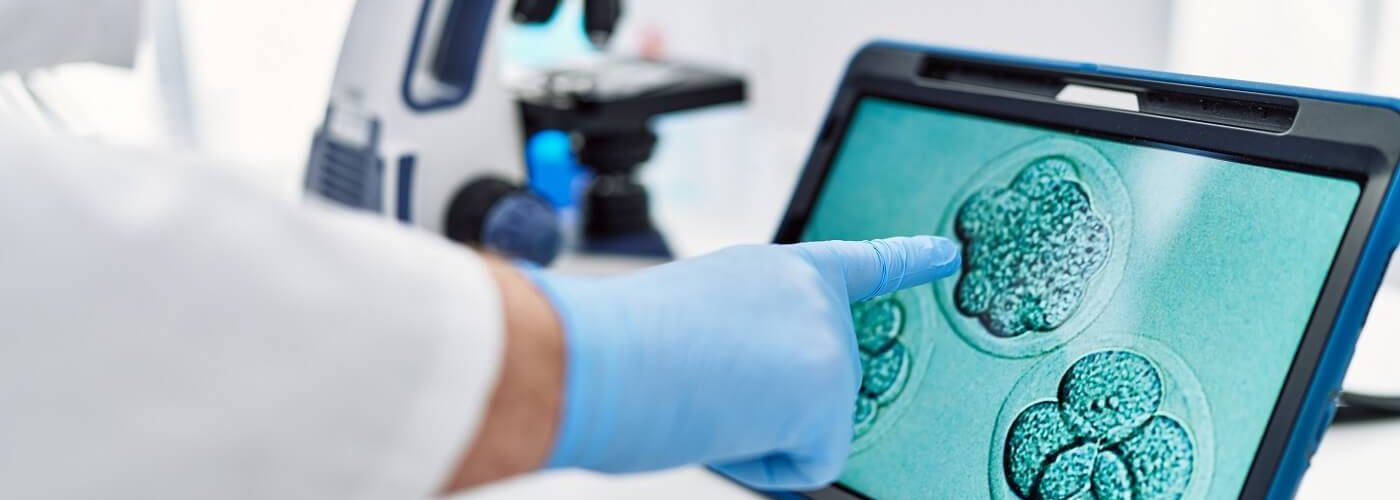
- At the 10th International IVIRMA Congress, the Weizmann Institute of Science presented its technique for generating synthetic mouse embryo models from stem cells grown only in a Petri dish, i.e., without fertilized eggs and without the need for a uterus.
- The researchers achieved mouse embryos with progenitor cells with a beating heart, a brain with well-formed folds or an incipient blood circulation (among other basic organs) in only eight days of development, reaching 95% similarity with natural embryos of these animals.
- In the future, synthetic embryo models could become a reliable source of cells, tissues, and organs for transplants, as well as helping to reduce the use of animals in research.
MALAGA, 24 APRIL 2023
The latest findings in assisted reproduction could be the key to the creation of tissues and organs that could be used in the future for human transplants. This has just been presented at the recently held 10th International IVIRMA Congress, in Malaga. Specifically, this team of researchers led by Dr. Jacob Hanna, from the Department of Molecular Genetics at the Weizmann Institute of Science, created synthetic mouse cells with no developmental restrictions and discovered a potential for embryonic and extraembryonic development in electronically controlled platforms that simulate a uterus, thus generating complete embryos with organs.
The result was a synthetic mouse embryo model with progenitor or specialized cells and a beating heart, a brain with well-formed folds, a yolk sac, a neural tube, an intestinal tract, a placenta, and an incipient blood circulation at only eight days of development, almost half of the 20 days of gestation required for a mouse.
As Dr. Hanna, associate professor at the Weizmann Institute of Science, explains: “The embryo is the perfect starting point for generating organs and the best 3D bioprinter, and that is the key to being able to create mechanisms that allow us to ensure that stem cells differentiate from specialized cells in the body or that they directly form entire organs. To date this has been very difficult to achieve, and the key to success has been unlocking the stem cells’ self-organizing coding potential.”
How did they achieve this? As a starting point, they built on previous advances in their lab, such as reprogramming stem cells and returning them to their earliest stage. In addition, they relied on the efficacy of a womb-like device to grow mouse embryos (natural embryos, in this previous research) using a nutrient solution inside continuously moving vessels, simulating the way nutrients are delivered by blood flow to the placenta and tightly controlling oxygen exchange and atmospheric pressure.
In the new study, the team set out to grow a synthetic embryo model solely from mouse stem cells that had been cultured for years in a Petri dish, avoiding the need to start from a fertilized egg. Before placing these cells in the ex-utero device, they divided them into 3 groups: one in which they were left as they were and two others that were pretreated to produce extraembryonic tissue. When mixed in the device, 0.5% formed spheres that developed into an embryo-like structure. Subsequently, the researchers were able to observe the placenta and yolk sacs forming outside the embryos and the development of the synthetic model in the same way as in a natural embryo.
“When compared with natural mouse embryos, the synthetic models showed 95% similarity in both the shape of the internal structures and the gene expression patterns of the different cell types. The organs observed in the models gave every indication of being functional,” said Dr. Hanna.
A range of possibilities for future transplants
The more realistic long-term goal is to study how stem cells form various organs in the developing embryo in order to open up new therapeutic horizons for organ transplants. This could lead to the possibility of one day being able to grow tissues and organs using synthetic embryo models.
But in order to have the possibility of developing cells for therapeutic purposes, it is necessary to understand their reprogramming and differentiation mechanisms, observing these transitions of stem cells during embryogenesis and organogenesis, as well as studying the degree of equivalence of cells in vitro with those in vivo.
In addition, this project could help to simplify the ethical debate around experimenting with natural embryos, in addition to reducing laboratory testing on animals. To a large extent, the technical and ethical problems posed by the use of natural embryos in research and biotechnology could be circumvented. Even in the case of mice, certain experiments are currently unfeasible because they would require thousands of embryos, whereas access to models derived from mouse embryonic cells, which grow in laboratory incubators by the millions, is virtually unlimited.
The next challenge is to understand how stem cells know what to do: how they self-assemble into organs and find their way to their assigned places within an embryo. And because, unlike a uterus, this system is transparent, it may prove useful for modeling birth and implantation defects in human embryos.
“Instead of developing a separate protocol to grow each cell type, e.g., kidney or liver cells, perhaps one day we can create a synthetic embryo-like model and then isolate the cells we need. We will not have to dictate to the emerging organs how they should develop. The actual embryo does it better,” concluded Dr. Hanna.
About the 10th International IVIRMA Congress
From 20 to 22 April, the 10th International IVIRMA Congress on Assisted Reproduction will be held, attracting the main researchers worldwide in the sector. At this event we present the advances achieved in the field of Reproductive Medicine, the most innovative techniques, and the results of the latest research. In addition, it is a meeting point for sharing best practices to improve the daily results in this activity. The Congress, the tenth edition of which is held this year in Malaga, brings together more than 1,200 specialists from 57 countries, and is held every two years.
